Abstract
Objective: Statins are commonly used in the management of dyslipidemia, with their efficacy varying based on type and dose. They are prescribed for both primary and secondary cardiovascular prevention. However, achieving recommended low-density lipoprotein cholesterol (LDL-C) targets remains a challenge in clinical practice. This study aimed to assess achievement of target LDL-C levels in statin users and to identify modifiable factors related to treatment failure.
Methods: This cross-sectional study was conducted in a family medicine outpatient clinic and included adults (≥18 years) on statin therapy for at least six months. Data were collected through face-to-face interviews using a sociodemographic questionnaire and the Adherence to Refills and Medications Scale (ARMS-7). Cardiovascular risk levels were classified using the SCORE risk calculator, and LDL-C goal attainment was assessed based on risk categories.
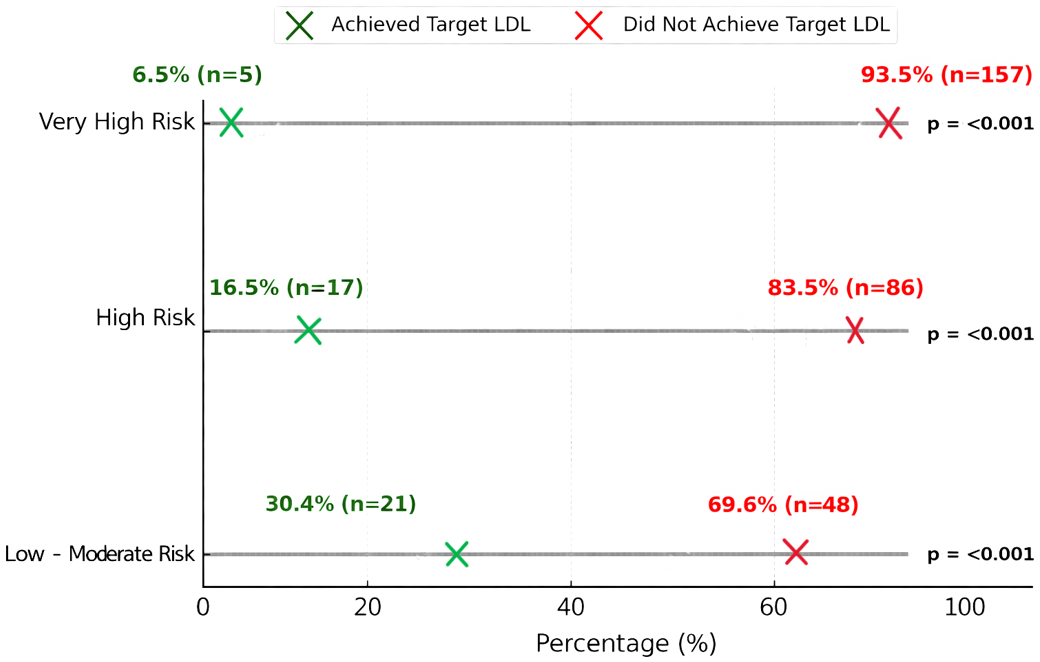
Results: Of 334 participants, 51.8% were male. LDL-C levels exceeded 100 mg/dL in 43.1% of patients. According to SCORE classifications, 55.4% were identified as being at very high cardiovascular risk. Overall, 87.1% of participants failed to achieve target LDL-C levels. Target LDL-C levels were achieved by 30.4% of participants in the low-to-moderate risk group, 16.5% in the high-risk group, and only 6.5% in the very high-risk group (p<0.001). In the study population, patients using statins solely for dyslipidemia (without a history of cardiovascular disease) and those with hypertension, were more likely to achieve LDL-C targets (p<0.05). A significantly higher proportion of male participants (16.8%) achieved the target LDL-C level compared to female participants (8.7%) (p<0.05).
Conclusion: The majority of patients on statin therapy did not meet their target LDL-C levels, particularly those at high cardiovascular risk, highlighting the impact of clinical inertia and underscoring the importance of individualized statin dosing and regular monitoring based on patients’ cardiovascular risk assessments in primary care settings.
Keywords: Primary health care, LDL cholesterol, treatment failure
Introduction
Dyslipidemia refers to abnormalities in plasma lipid levels that elevate cardiovascular risk, including increased total cholesterol, low-density lipoprotein cholesterol (LDL-C), and triglycerides, as well as decreased High-Density Lipoprotein Cholesterol (HDL-C) or hypoalphalipoproteinemia. These lipid disturbances are major contributors to the development of cardiovascular diseases, which remain the leading cause of mortality worldwide.[1] Achieving guideline-recommended LDL-C targets remains a major challenge worldwide, particularly in high cardiovascular risk patients. For instance, the DA VINCI study reported that only 44% of high-risk patients across various European regions reached the 2016 ESC/EAS LDL-C targets.[2] In the TEKHARF 2011/14 cohort, LDL-C >130 mg/dL was observed in 39% of men and in 46% of women in a sample predominantly composed of postmenopausal women.[3]
However, data on LDL-C target attainment among patients receiving statin therapy in routine primary care are limited. This evidence gap highlights the need to evaluate LDL-C goal achievement in the Turkish population to optimize treatment strategies and reduce cardiovascular risk. According to established guidelines, the term dyslipidemia refers to deviations from target lipid values.[4] From a cardiovascular perspective, increases in total cholesterol and LDL-C, along with decreases in HDL-C, are defined as dyslipidemia.[5] Lowering LDL-C levels is currently the most crucial strategy in combating atherosclerotic diseases.[6]
Beyond LDL-C, non-HDL-C and apolipoprotein B have been shown to be stronger predictors in the assessment of cardiovascular risk.[7] In addition, elevated levels of lipoprotein(a) confer genetically determined additional atherogenic risk and are increasingly recognized as an important component of the residual cardiovascular risk under current treatment strategies.[8]
The effects of statins on the lipid profile are dose-dependent and vary according to the type of statin used. In high-intensity statin therapy, the goal is to achieve a ≥50% reduction in LDL-C levels. In moderate-intensity statin therapy, the target is a 30–50% reduction in LDL-C. Statins reduce triglyceride levels by approximately 10–20% on average. [9,10] Their effects on HDL-C are independent of their impact on LDL-C and are typically associated with a 1–10% increase.[11] In the treatment of dyslipidemia, the target lipid levels are determined based on cardiovascular risk stratification. Non-statin options include proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors, colesevelam, ezetimibe, fibrates, and niacin, used alone or in combination.[12]
The cornerstone of dyslipidemia treatment is the use of lipid-lowering medications to achieve optimal lipid control and reduce cardiovascular disease morbidity and mortality.[13] However, despite the widespread availability and prescription of these highly effective agents, a significant proportion of patients fail to reach recommended lipid targets. This gap highlights the critical role of treatment adherence in the effective management of dyslipidemia and its impact on overall therapeutic outcomes. Statin use and dose optimization can be limited in practice due to statin intolerance, with muscle symptoms reported by approximately 60% of adults as the main reason for discontinuation, which in turn affects achieving target levels.[14,15]
This study evaluates LDL-C goal attainment in routine primary care, highlighting real-world usage and patient adherence. In addition, medication adherence was quantitatively assessed using the ARMS 7 scale and analyzed alongside lipid outcomes. The combination of a large, real-world sample with integrated adherence and risk assessment provides a comprehensive perspective on factors influencing LDL-C target achievement. This underscores the need for individualized statin therapy and goal-oriented monitoring, offering direct implications for clinical practice. This study aimed to examine the rates of achieving target LDL-C levels among patients using statins and to identify the contributing factors, to identify modifiable factors associated with treatment failure, which can inform targeted interventions in primary care.
Materials and Methods
Our study has a descriptive and cross-sectional design. A total of 334 volunteers who applied to the Family Medicine Outpatient Clinic of Eskişehir Osmangazi University Hospital between April 1, 2024, and September 1, 2024, were included in the study. The minimum sample size of 285 was calculated using OpenEpi (5% margin of error, 50% frequency, 97% confidence level). The inclusion criteria were being over 18 years of age and receiving statin therapy for at least six months. Patients were excluded if they had familial hypercholesterolemia, a history of acute coronary syndrome within the previous three months, severe hepatic or renal disease, active malignancy, or were pregnant or breastfeeding. Cases with missing data were excluded from the analysis. The study was conducted in accordance with the Declaration of Helsinki and approved by the Non-invasive Clinical Research Ethics Committee of Eskişehir Osmangazi University (Protocol No: 16, Approval Date: March 19, 2024). Informed consent was obtained from all participants.
Data collection methods
After obtaining written informed consent from the participants, data were collected through face-to-face interviews using a structured questionnaire that included sociodemographic characteristics and certain health assessments, as well as the Medication Adherence and Prescription Refill Scale-7. The sociodemographic questionnaire included questions regarding smoking and alcohol use, marital status, existing chronic conditions, medications used, physical activity status, dietary adherence, and whether they had heard any negative comments about statins. Chronic conditions were identified through a combination of self-reported diagnoses and supporting clinical measurements (e.g., blood pressure readings, medication lists) when available. Blood pressure was measured using a validated sphygmomanometer following standard protocols, with patients in a seated position after a rest period, at least 5 days per week, preferably every day. Physical activity status and dietary adherence were assessed using single-item, self-reported questions. Participants were asked whether they engaged in regular physical activity and whether they generally adhered to a healthy diet.
The patient’s laboratory data were obtained from the hospital records, and the most recent values at the time of admission were recorded.
Adherence to Refills and Medications Scale-7 (ARMS‐7)
The scale consists of 7 items and is based on a 4-point Likert-type format. Items 1, 2, 3, and 6 assess medication adherence, while items 4, 5, and 7 evaluate prescription refill behavior. Each item is scored from 1 to 4, where 1=never, 2=sometimes, 3=often, and 4=always. The total score ranges from 7 to 28. Higher scores on the scale indicate poorer medication adherence. The validity and reliability study of the scale in Türkiye was conducted by Gökdoğan F. and the Cronbach’s alpha coefficient was found to be 0.75.[16,17]
Determination of target LDL-C levels
Participants were classified into cardiovascular risk categories (very high, high, moderate, and low risk) according to the recommendations of the 2021 TEMD Dyslipidemia Guideline and the 2019 ESC Dyslipidemia Guidelines.[5,12] Target LDL-C levels were determined based on these cardiovascular risk categories. In addition, while categorizing patients’ cardiovascular risk, we used the SCORE Risk Calculator, as recommended by the same guidelines for high-risk countries.[18] The target LDL-C levels in treatment were defined as follows: <55 mg/dL for the very high-risk group, <70 mg/dL for the high-risk group, and <100 mg/dL for the moderate-risk group.[5]
Statistical analysis
Data were analyzed using IBM SPSS Statistics for Windows, Version 23.0 (IBM Corp., Armonk, NY, USA). In descriptive statistics, categorical variables are presented as frequencies and percentages, while continuous variables are expressed as mean, standard deviation, median, minimum, and maximum values (reported as the range) The normality of continuous variables was assessed using the Shapiro-Wilk test. Since normal distribution assumptions were not met, the Mann-Whitney U test was used to compare two means. Pearson Chi-Square test was used for comparing categorical variables. A confidence interval of 95% and a significance level of p<0.05 were accepted for all statistical analyses.
Results
Of the participants, 48.2% (n=161) were female and 51.8% (n=173) were male. The mean age was 63.0±9.5 years, with a minimum of 40 and a maximum of 85 years.
A greater proportion of male participants (16.8%) achieved the target LDL-C level compared to female participants (8.7%). The difference in the achievement of target LDL-C levels between males and females was statistically significant (p=0.028). However, there was no statistically significant difference in the mean age between participants who achieved the target LDL-C level and those who did not (p=0.311) (Table 1).
| * Mann-Whitney U test | ||||
| Table 1. The relationship between participants' sociodemographic characteristics and achievement of target LDL-C levels. | ||||
| Sociodemographic Characteristics |
|
|
|
|
|
|
|
|||
| Age (Mean ± SD) |
|
|
|
|
| Gender |
|
|
||
| Female |
|
|
|
|
| Male |
|
|
||
| Marital Status | ||||
| Married |
|
|
|
|
| Single |
|
|
||
| BMI | ||||
| Normal |
|
|
|
|
| Overweight |
|
|
||
| Obese |
|
|
||
| Alcohol Use | ||||
| Yes |
|
|
|
|
| No |
|
|
||
| Smoking | ||||
| Yes |
|
|
|
|
| No |
|
|
||
In the analysis of the LDL-C levels of the participants, it was found that 43.1% (n=144) had LDL-C levels above 100 mg/dL, 37.7% (n=126) were in the 71-100 mg/dL range, 14.1% (n=47) were in the 55-70 mg/dL range, and 5.1% (n=17) had LDL-C levels below 55 mg/dL. The participants' lipid profile showed a mean total cholesterol of 179±51 mg/dL (median 169, range 88–424), triglycerides 165±108 mg/dL (median 140, range 38–1308), LDL-C 104±41 mg/dL (median 95, range 27–298), and HDL-C 46±12 mg/dL (median 45, range 10–87).
According to cardiovascular risk categories; 13.8% of participants (n=46) were found to be at low-medium risk, 30.8% (n=103) at high risk, and 55.4% (n=185) at very high risk.
Based on cardiovascular risk categories, 12.9% of the participants (n=43) achieved the target LDL-C level, whereas 87.1% (n=291) failed to reach the target level.
The attainment of target LDL-C levels varied significantly across cardiovascular risk categories. Participants in the low–moderate risk group achieved target LDL-C levels at a rate of 30.4%, compared to 16.5% in the high-risk group and only 6.5% in the very high-risk group (p<0.001). This trend is visually depicted in Figure 1, where a marked decline in LDL-C goal attainment is observed with increasing cardiovascular risk.
The mean systolic blood pressure of the participants is 120.0±11.8 mmHg, and the mean diastolic blood pressure is 72.7±7.6 mmHg. There is no statistically significant relationship between systolic blood pressure and achievement of target LDL-C levels (p=0.372). Similarly, there is no statistically significant relationship between diastolic blood pressure and achievement of target LDL-C levels (p=0.201).
25.5% (n=85) of the participants were found to be normal, 42.8% (n=143) were overweight, and 31.7% (n=106) were obese. There is no statistically significant relationship between body mass index and reaching the target LDL-C value (p=0.870).
In the analysis the reasons for participants' initiation of statin therapy, it was found that 52.1% (n=174) started treatment due to dyslipidemia, 37.4% (n=125) due to coronary artery disease, 9.9% (n=33) due to myocardial infarction, and 0.6% (n=2) due to angina pectoris. In the analysis the achievement of target LDL-C levels based on the reasons for statin use, participants using statins due to dyslipidemia had a higher rate of reaching the target LDL-C level compared to those using statins for cardiac reasons (p=0.013) (Table 2).
| * Pearson Chi-Square test | ||||
| Table 2. The relationship between reason for statin use and achievement of target LDL-C levels. | ||||
| Reason for Statin Use |
|
|
|
|
|
|
|
|||
| Dyslipidemia |
|
|
|
|
| Cardiac Reasons |
|
|
||
20.9% of participants have been using statins for 6 months to 1 year, 23.6% for 1-3 years, 17.4% for 3-5 years, 19.8% for 5-10 years, 8.7% for 10-15 years, 3.6% for 15-20 years, and 6.0% for more than 20 years. There is no statistically significant relationship between the duration of statin use and achievement of target LDL-C levels (p=0.139).
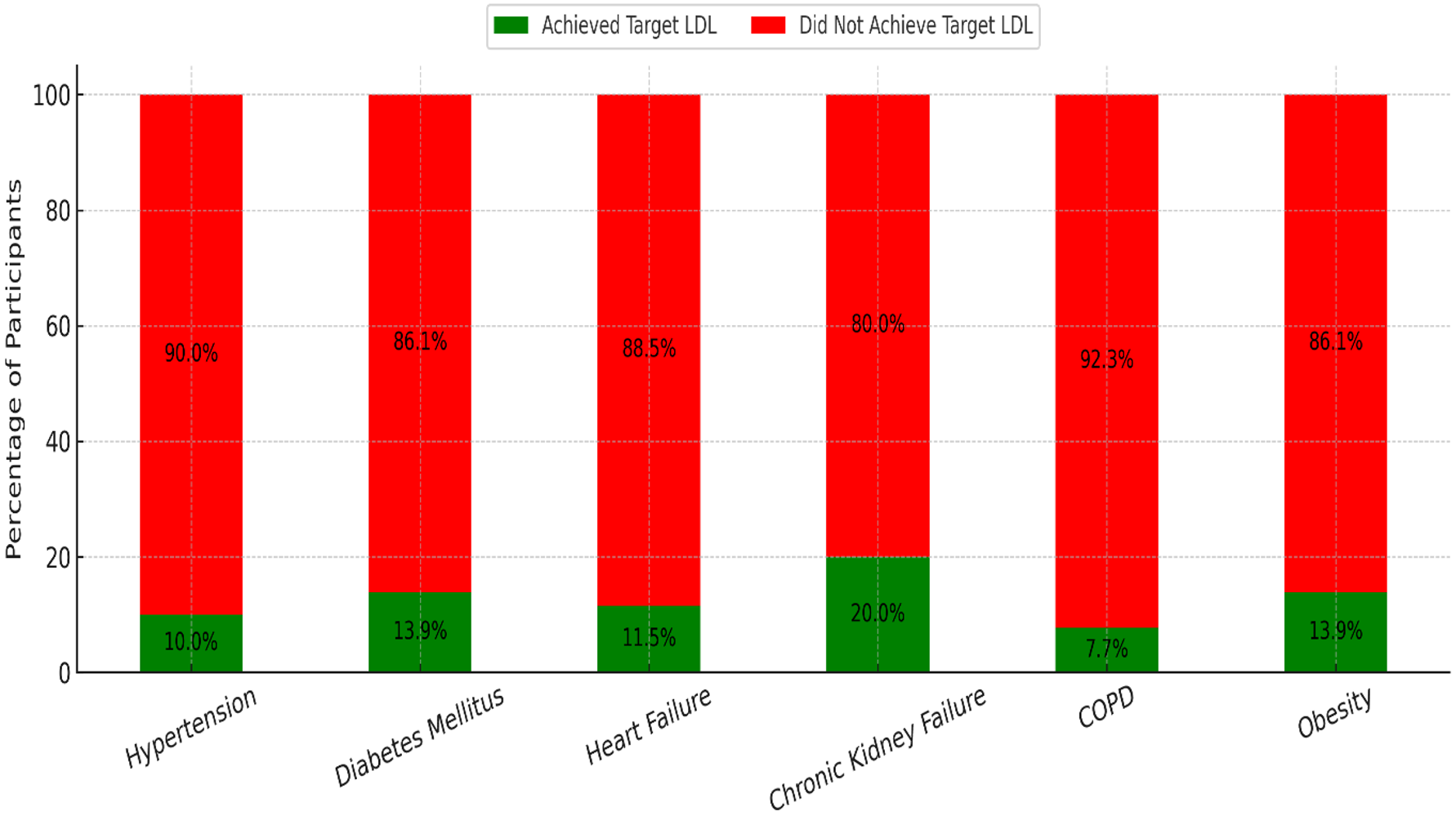
71.9% (n=240) of participants have hypertension, 56.0% (n=187) have diabetes mellitus, 15.6% (n=52) have heart failure, 12.0% (n=40) have chronic kidney failure, and 7.8% (n=26) have COPD. Figure 2 illustrates the proportion of participants achieving target LDL-C levels according to the presence or absence of various chronic diseases. Hypertension was the only condition significantly associated with lower target LDL-C attainment (p=0.012). Participants with hypertension achieved target LDL-C levels at a rate of 10.0%, compared to 20.2% in those without hypertension. Other chronic conditions, including diabetes mellitus, heart failure, chronic kidney failure, COPD, and obesity, did not show statistically significant differences in LDL-C target attainment rates (Figure 2).
In the analysis of the types and dosages of statins used by participants, it was found that the most commonly used statin was atorvastatin 20 mg/day (41.0%), followed by atorvastatin 10 mg/day (29.0%) and atorvastatin 40 mg/day (18.6%). There was no statistically significant relationship between the type and dosage of statins used by participants and the achievement of target LDL-C levels (p>0.05).
94.9% of participants (n=317) did not use any additional lipid-lowering medications, while 2.7% (n=9) used fibrates, 1.8% (n=6) used ezetimibe, and 0.6% (n=2) used niacin.
There was no statistically significant relationship between physical activity and achievement of target LDL-C levels (p=0.374).
The mean score on the Adherence to Refills and Medications Scale-7 (ARMS-7) was 8.9±4.7. The mean score for the Medication Adherence subscale was 5.0±2.6, and the mean score for the Prescription Refill subscale was 3.9±2.1.
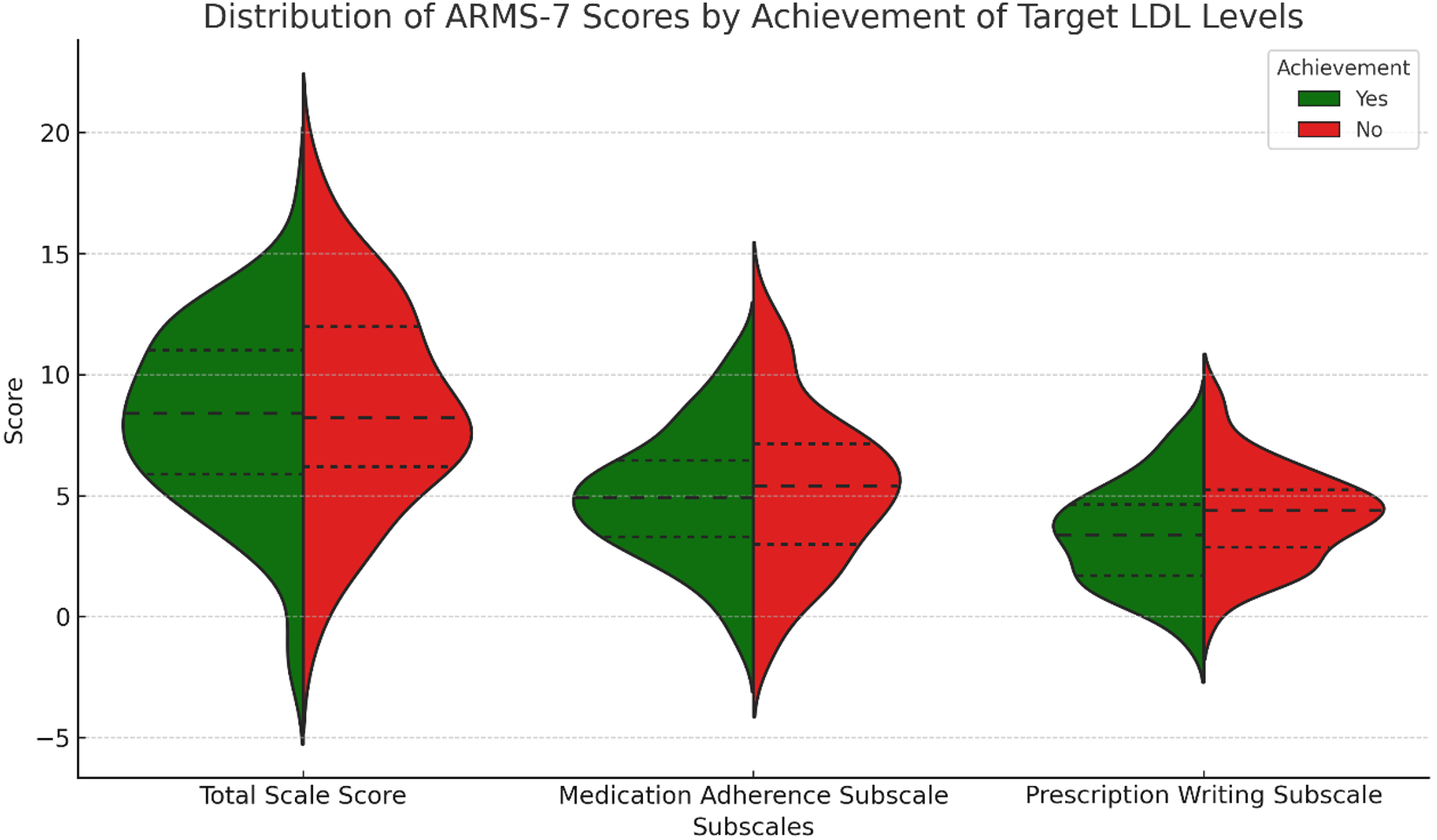
The mean ARMS-7 total scale score was slightly lower among participants who achieved target LDL-C levels (8.4 ± 4.2) compared to those who did not (9.0 ± 4.7). Similarly, mean scores on the Medication Adherence subscale (4.7 ± 2.4 vs. 5.0 ± 2.7) and Prescription Refill subscale (3.7 ± 1.9 vs. 3.9 ± 2.1) were marginally lower in the LDL-C-achieving group. However, these differences did not reach statistical significance (p>0.05 for all comparisons). Although not statistically significant, slightly lower ARMS-7 scores in participants achieving target LDL-C suggest better medication adherence may contribute to improved lipid control.
Figure 3 displays violin plots of ARMS-7 total and subscale scores by achievement of target LDL-C levels. The total score distribution was higher among patients who did not achieve target LDL-C, whereas the distributions of the medication adherence and prescription refill subscales showed no statistically significant differences between the groups (p>0.05) (Figure 3).
Discussion
A large proportion of patients receiving statin therapy failed to achieve their target LDL-C levels (87.1%), with attainment rates being lowest among those at very high cardiovascular risk (6.5%). Male participants were more likely to reach LDL-C goals compared to females, and target achievement was higher among individuals with hypertension and those using statins solely for dyslipidemia without established cardiovascular disease. These findings indicate a significant treatment gap in the management of dyslipidemia within primary care and emphasize the need for more personalized treatment strategies, including appropriate dose adjustment, close monitoring, and enhanced adherence support. Overall, the results highlight the pivotal role of family physicians in optimizing lipid-lowering therapy and improving cardiovascular prevention outcomes.
When cardiovascular risk calculations were evaluated in our study, only 12.9% of participants achieved their target LDL-C levels. Similarly, the CEPHEUS (Centralized Pan-European Survey on the Undertreatment of Hypercholesterolemia) study, which included 575 dyslipidemic patients from nine centers in Türkiye, also reported low rates of LDL-C target attainment.[19] According to the Adult Treatment Panel III (ATP III) guidelines of the National Cholesterol Education Program (NCEP) (2004), the proportion of patients achieving target LDL-C levels was 35.1%.[20] The findings of our study are consistent with national data, indicating persistent challenges in the implementation of guideline-based dyslipidemia management. These results suggest that treatment strategies and patient adherence require closer evaluation and optimization to improve lipid control outcomes.
It was observed that 43.1% of participants had LDL-C levels above 100 mg/dL. In many comprehensive studies conducted in our country, the average LDL-C level is above 100 mg/dL.[21,22] In a systematic review by Kızılırmak P et al have shown that with treatment, 47.9% of patients had LDL-C levels reduced to below 100 mg/dL. After low-dose treatment, this rate was found to be 41.0%, and after high-dose statin treatment, it was 66.1%.[23] The low use of high-intensity statins and combination therapies in our study partly explains this finding; however, other factors such as diet, physical activity, and family predisposition also play a role.
These data indicate that the targets proposed by the guidelines have not been sufficiently reflected in clinical practice.
Several studies have also examined the relationship between statin intensity and target LDL-C attainment. According to the 2019 ESC Guidelines, high-intensity statin therapy at the maximum tolerated dose is recommended to achieve LDL-C targets.[5] In our study population, although 23.7% of patients received high-intensity statin therapy, only 12.9% reached their target LDL-C levels. Failure to achieve LDL-C targets in patients receiving high-intensity statins may be related to poor adherence, statin-related side effects, and individual cardiovascular risk profiles.[24] Moreover, while high-intensity statin therapy reduces cardiovascular risk, it does not always guarantee LDL-C target attainment.[25,26] Collectively, these results highlight the need for individualized statin dosing, regular follow-up, and adherence strategies in primary care to improve lipid control and cardiovascular outcomes.
Many studies in the literature have reported that longer durations of statin therapy are associated with a reduction in cardiovascular events, which has been attributed to improved treatment adherence over time and the more pronounced long-term effects of the medication.[26,27] In our study, 94.9% of participants were not receiving any additional lipid-lowering agents alongside statins, and the rate of fibrate use was only 2.7%. Similar patterns have been observed in large-scale studies conducted in Türkiye, where the low utilization of non-statin lipid-lowering medications limits the ability to assess their overall impact. In the CEPHEUS study, despite low rates of LDL-C target attainment, 70.5% of patients did not have any changes to their initial lipid-lowering therapy or dosage adjustments.[19]
As patients' cardiovascular risk increases, a significant decrease is observed in the rates of achieving target LDL-C levels. In high-risk groups, the target LDL-C level is set lower, which contributes to a lower attainment rate. In contrast, in low-risk groups, the target LDL-C level is more attainable. Especially in high and very high-risk patients, high-intensity statins should be preferred, and in cases where monotherapy is insufficient, it may be necessary to add Ezetimibe or PCSK9 inhibitors to the treatment regimen.[28]
The rate of achieving target LDL-C levels was 8.7% in women, compared to 16.7% in men. Women had a significantly lower rate of target attainment than men. According to the literature, it is well known that the burden of cardiovascular and metabolic diseases increases in postmenopausal women due to the decline in estrogen levels. In addition, factors such as increased systemic inflammation, HDL-C dysfunction, and gene–environment interactions contribute to the more adverse outcomes observed in women. Moreover, it has been shown that the absorption of long-chain fatty acids is higher in women.[29] In our study, the rate of achieving target LDL-C levels was significantly lower in patients diagnosed with hypertension compared to those without hypertension. The coexistence of hypercholesterolemia and hypertension in adults, and its contribution to cardiovascular disease risk, has been well documented. Previous studies report that approximately one in ten Turkish adults over the age of 30 has elevated LDL-C levels in combination with hypertension, with an age- and sex-adjusted relative risk for CVD increased by 2.4 times.[30] However, the lower achievement rate observed in our hypertensive cohort should be interpreted with careful consideration of clinical context. Patients with hypertension are frequently categorized as very high-risk according to contemporary guidelines, which subjects them to a much stricter LDL-C target (<55 mg/dL). Therefore, the lower success rate in this group may not solely reflect the presence of hypertension itself, but rather the difficulty of attaining this more demanding therapeutic threshold. In this regard, the observed “failure” likely represents the consequences of a more stringent LDL-C goal rather than a direct effect of hypertension.[5]
The low LDL-C target attainment in hypertensive patients may, in part, reflect “clinical inertia,” where treatment intensification is often delayed in patients with multiple comorbidities despite unmet goals. Therapeutic inertia, sometimes referred to as clinical inertia, has been defined as failure to initiate or intensify therapy when therapeutic goals are not reached. It is relevant for conditions where therapeutic targets are clearly defined, the benefits of reaching those targets are well established, effective therapies are accessible, and up-to-date clinical guidelines are available.[31,32]
Recognizing this phenomenon underscores the importance of proactive, risk-based management strategies to improve lipid control in this high-risk population. Furthermore, in individuals with chronic conditions such as hypertension and diabetes, adherence to statin therapy tends to be more difficult, and side effects are more frequently observed, which in turn makes achieving target LDL-C levels more challenging.[29]
Obesity is a well-established major risk factor for the development of cardiovascular disease. In our study, obesity was present in 30.2% of the patient population, highlighting its significant role in the control of LDL-C levels. The inability to achieve effective weight management in these patients may have contributed to the failure to reach target LDL-C levels.
In addition to pharmacological treatment, adopting a healthy lifestyle is an essential component of dyslipidemia management. In our study, only 17.7% of participants reported engaging in regular exercise 2–3 times per week. According to the literature, the combination of statin therapy and exercise has been shown to significantly reduce LDL-C levels.[33] The low number of patients engaging in regular physical activity in our cohort may have negatively influenced the rate of achieving target LDL-C levels. Therefore, at every clinical visit, patients should be reminded of the importance of healthy lifestyle changes and provided with accessible exercise programs.
In the ARMS-7 survey conducted to assess medication adherence and prescription behavior, the mean adherence score among patients who achieved target LDL-C levels was 4.7 ± 2.4, while the mean prescription score was 3.7 ± 1.9. These findings are consistent with previous national data and align with international reports indicating suboptimal LDL-C target attainment despite adequate self-reported adherence.[34-36] Nevertheless, failure to achieve LDL-C targets may be explained by the lack of regular LDL-C monitoring during follow-up visits and insufficient statin dose titration. These results highlight the role of patient behavior in clinical outcomes and provide a foundation for designing future health education and follow-up strategies. However, considerable variability observed within groups suggests that factors beyond self-reported adherence—such as patient clinical characteristics or treatment-related factors—may have a greater influence on LDL-C goal attainment.
Recent studies have similarly emphasized that therapeutic inertia, including limited treatment intensification or inadequate statin dose adjustment, remains a major barrier to achieving LDL-C targets, even among patients reporting good adherence.[37-39] For instance, in the Turkish AIZANOI study, only 26.2% of high- and very high-risk patients achieved target LDL-C levels, largely due to physician-related inertia.[35] Likewise, Bayram et al. (2020) reported low statin utilization and poor LDL-C target attainment among patients with type 2 diabetes.[34] Collectively, these findings suggest that while patient adherence is an important determinant, system-level barriers and clinician-related factors play a decisive role in lipid management outcomes. Future research should examine the interplay between patient behavior and health system processes, and evaluate interventions targeting both adherence support and clinical decision-making. Clinician-related factors contributing to therapeutic inertia include gaps in knowledge, uncertainty about diagnosis, treatment goals, or evidence, concerns about the safety of intensifying therapy, limited time, and lack of access to or integration of clinical guidelines and decision support. Additional contributors are insufficient system support for structured follow-up to review goal attainment and the absence of a team-based care approach involving pharmacists.[40]
In this study, 87.1% of patients failed to achieve their risk-based LDL-C targets. This proportion is consistent with findings from large-scale European data such as the DA VINCI study highlighting a substantial treatment gap in primary care.[2] Our results further demonstrate that as cardiovascular risk increases, the likelihood of achieving LDL-C goals decreases, underscoring the challenge of meeting more stringent targets in high-risk groups.[5] Notably, patients prescribed statins for “dyslipidemia” achieved LDL-C goals more frequently than those prescribed for “cardiac reasons,” which can be explained by the stricter LDL-C targets in the secondary prevention population. This finding reflects an important nuance in how risk stratification influences treatment success. Overall, our study provides quantitative confirmation of the treatment gap in Türkiye and emphasizes the need for strengthened lipid-lowering strategies in routine practice.
Limitations
A limitation of this study is the potentially insufficient statistical power in certain subgroups, which may explain nonsignificant findings. Unmeasured potential confounders, such as detailed dietary intake, may have influenced LDL-C target attainment. In addition, the single-center design may limit the generalizability of the findings to other primary care settings in Türkiye. Repeating the study with patient randomization and a larger sample size will provide more definitive information and enhance the validity of the results.
Conclusions
Our study highlights the presence of clinical inertia in lipid management within primary care in Türkiye. Despite clear guideline-based LDL-C targets, 87.1% of patients failed to achieve their risk-based goals, with the lowest attainment observed in very high-risk groups. Most patients were treated with low- to moderate-intensity statins, and dose titration or combination therapies were infrequently utilized, reflecting a lack of treatment intensification. Additionally, lower target attainment among women, hypertensive, and obese patients suggests that clinicians may face challenges in managing more complex or high-risk populations. Limited structured follow-up, insufficient system support, and the absence of a team-based care approach further exacerbate the gap between risk stratification and therapeutic action. These findings quantitatively demonstrate how clinician- and system-related factors contribute to clinical inertia, emphasizing the need for proactive, individualized treatment strategies to optimize LDL-C control and reduce cardiovascular risk.
Ethical approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Non-invasive Clinical Research Ethics Committee of Eskişehir Osmangazi University (protocol code 16 and date of approval March 19, 2024).
Source of funding
The authors declare that the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest to disclose.
References
- Ballena-Caicedo J, Zuzunaga-Montoya FE, Loayza-Castro JA, et al. Global prevalence of dyslipidemias in the general adult population: a systematic review and meta-analysis. J Health Popul Nutr. 2025;44(1):308. https://doi.org/10.1186/s41043-025-01054-3
- Ray KK, Molemans B, Schoonen WM, et al. EU-Wide Cross-Sectional Observational Study of Lipid-Modifying Therapy Use in Secondary and Primary Care: the DA VINCI study. Eur J Prev Cardiol. 2021;28(11):1279-1289. https://doi.org/10.1093/eurjpc/zwaa047
- Onat A, Karakoyun S, Akbaş T, et al. Turkish Adult Risk Factor survey 2014: overall mortality and coronary disease incidence in Turkey's geographic regions. Turk Kardiyol Dern Ars. 2015;43(4):326-332. https://doi.org/10.5543/tkda.2015.80468
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J Am Coll Cardiol. 2019;73(24):e285-e350. https://doi.org/10.1016/j.jacc.2018.11.003
- Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41(1):111-188. https://doi.org/10.1093/eurheartj/ehz455
- Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38(32):2459-2472. https://doi.org/10.1093/eurheartj/ehx144
- Ahmad M, Sniderman AD, Hegele RA. Apolipoprotein B in cardiovascular risk assessment. CMAJ. 2023;195(33). https://doi.org/10.1503/cmaj.230048
- Bashir B, Schofield J, Downie P, et al. Beyond LDL-C: unravelling the residual atherosclerotic cardiovascular disease risk landscape-focus on hypertriglyceridaemia. Front Cardiovasc Med. 2024;11:1389106. https://doi.org/10.3389/fcvm.2024.1389106
- Reiner Z. Managing the residual cardiovascular disease risk associated with HDL-cholesterol and triglycerides in statin-treated patients: a clinical update. Nutr Metab Cardiovasc Dis. 2013;23(9):799-807. https://doi.org/10.1016/j.numecd.2013.05.002
- Sharma A, Joshi PH, Rinehart S, Thakker KM, Lele A, Voros S. Baseline very low-density lipoprotein cholesterol is associated with the magnitude of triglyceride lowering on statins, fenofibric acid, or their combination in patients with mixed dyslipidemia. J Cardiovasc Transl Res. 2014;7(4):465-474. https://doi.org/10.1007/s12265-014-9559-3
- Barter PJ, Brandrup-Wognsen G, Palmer MK, Nicholls SJ. Effect of statins on HDL-C: a complex process unrelated to changes in LDL-C: analysis of the VOYAGER Database. J Lipid Res. 2010;51(6):1546-1553. https://doi.org/10.1194/jlr.P002816
- Turkish Endocrinology and Metabolism Association. Dyslipidemia diagnosis and treatment guide 2021. Available at: https://file.temd.org.tr/Uploads/publications/guides/documents/20211026164301-2021tbl_kilavuzb66456ad2f.pdf
- Berman AN, Blankstein R. Optimizing dyslipidemia management for the prevention of cardiovascular disease: a focus on risk assessment and therapeutic options. Curr Cardiol Rep. 2019;21(9):110. https://doi.org/10.1007/s11886-019-1175-z
- Bytyçi I, Penson PE, Mikhailidis DP, et al. Prevalence of statin intolerance: a meta-analysis. Eur Heart J. 2022;43(34):3213-3223. https://doi.org/10.1093/eurheartj/ehac015
- Jacobson TA, Cheeley MK, Jones PH, et al. The statin adverse treatment experience survey: experience of patients reporting side effects of statin therapy. J Clin Lipidol. 2019;13(3):415-424. https://doi.org/10.1016/j.jacl.2019.04.011
- Gökdoğan F, Kes D. Validity and reliability of the Turkish adherence to refills and medications scale. Int J Nurs Pract. 2017;23(5):10.1111/ijn.12566. https://doi.org/10.1111/ijn.12566
- Kripalani S, Risser J, Gatti ME, Jacobson TA. Development and evaluation of the Adherence to Refills and Medications Scale (ARMS) among low-literacy patients with chronic disease. Value Health. 2009;12(1):118-123. https://doi.org/10.1111/j.1524-4733.2008.00400.x
- Conroy RM, Pyörälä K, Fitzgerald AP, et al. Estimation of ten-year risk of fatal cardiovascular disease in Europe: the SCORE project. Eur Heart J. 2003;24(11):987-1003. https://doi.org/10.1016/s0195-668x(03)00114-3
- Kültürsay H. Results of the rosuvastatin studies in Turkey. Arch Turk Soc Cardiol. 2007;35(1):24-30.
- Grundy SM, Cleeman JI, Merz CNB, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J Am Coll Cardiol. 2004;44(3):720-732. https://doi.org/10.1016/j.jacc.2004.07.001
- Onat A, Senocak MS, Surdum-Avci G, Ornek E. Prevalence of coronary heart disease in Turkish adults. Int J Cardiol. 1993;39(1):23–31. https://doi.org/10.1016/0167-5273(93)90293-p
- Uzunlulu M, Oğuz A, Tigen K. High-density lipoprotein cholesterol in coronary artery disease patients: is it as low as expected? Anadolu Kardiyol Derg. 2005;5(4):268-270.
- Kızılırmak P, Öngen Z, Kayıkçıoğlu M, Tokgözoğlu L. Evaluation of statin use on LDL cholesterol levels in Turkey: a systematic review. Turk Kardiyol Dern Ars. 2020;48(2):137-148. https://doi.org/10.5543/tkda.2019.45156
- Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet. 2005;366(9493):1267-1278. https://doi.org/10.1016/S0140-6736(05)67394-1
- Ridker PM, Danielson E, Fonseca FAH, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359(21):2195-2207. https://doi.org/10.1056/NEJMoa0807646
- Yusuf S, Bosch J, Dagenais G, et al. Cholesterol lowering in ıntermediate-risk persons without cardiovascular disease. N Engl J Med. 2016;374(21):2021-2031. https://doi.org/10.1056/NEJMoa1600176
- Strandberg TE, Kovanen PT, Lloyd-Jones DM, et al. Drugs for dyslipidaemia: the legacy effect of the Scandinavian Simvastatin Survival Study (4S). The Lancet. 2024;404(10470):2462–2475. https://doi.org/10.1016/S0140-6736(24)02089-0
- Sabatine MS, Giugliano RP, Wiviott SD, et al. Efficacy and safety of evolocumab in reducing lipids and cardiovascular events. N Engl J Med. 2015;372(16):1500-1509. https://doi.org/10.1056/NEJMoa1500858
- Rosano GMC, Spoletini I, Vitale C. Cardiovascular disease in women, is it different to men? The role of sex hormones. Climacteric. 2017;20(2):125-128. https://doi.org/10.1080/13697137.2017.1291780
- Onat A, Karadeniz Y, Tusun E, Yüksel H, Kaya A. Advances in understanding gender difference in cardiometabolic disease risk. Expert Rev Cardiovasc Ther. 2016;14(4):513-523. https://doi.org/10.1586/14779072.2016.1150782
- Lebeau JP, Cadwallader JS, Aubin-Auger I, et al. The concept and definition of therapeutic inertia in hypertension in primary care: a qualitative systematic review. BMC Fam Pract. 2014;15:130. https://doi.org/10.1186/1471-2296-15-130
- Khunti K, Gomes MB, Pocock S, et al. Therapeutic inertia in the treatment of hyperglycaemia in patients with type 2 diabetes: a systematic review. Diabetes Obes Metab. 2018;20(2):427-437. https://doi.org/10.1111/dom.13088
- Costa RR, Vieira AF, Coconcelli L, et al. Statin use improves cardiometabolic protection promoted by physical training in an aquatic environment: a randomized clinical trial. Arq Bras Cardiol. 2021;117(2):270-278. https://doi.org/10.36660/abc.20200197
- Bayram F, Sonmez A, Haymana C, et al. Utilization of statins and LDL-cholesterol target attainment in Turkish patients with type 2 diabetes - a nationwide cross-sectional study (TEMD dyslipidemia study). Lipids Health Dis. 2020;19(1):237. https://doi.org/10.1186/s12944-020-01408-2
- Şen T, Dinç Asarcıklı L, Güven S, et al. Adherence to current dyslipidemia guideline in patients utilizing statins according to risk groups and gender differences: the AIZANOI study. Anatol J Cardiol. 2024;28(6):273-282. https://doi.org/10.14744/AnatolJCardiol.2024.4218
- Banach M, Reiner Ž, Surma S, et al. 2024 Recommendations on the optimal use of lipid-lowering therapy in established atherosclerotic cardiovascular disease and following acute coronary syndromes: a position paper of the International Lipid Expert Panel (ILEP). Drugs. 2024;84(12):1541-1577. https://doi.org/10.1007/s40265-024-02105-5
- Seo YH, Hong JY, Kim YK, Kim KH, Kwon TG, Bae JH. Therapeutic inertia in statin therapy for secondary prevention after percutaneous coronary intervention: a nationwide population-based cohort study. BMC Cardiovasc Disord. 2025;25(1):595. https://doi.org/10.1186/s12872-025-05081-0
- Cosín Sales J, Escobar Cervantes C, Gómez-Barrado JJ, et al. Impact of therapeutic inertia in lipid-lowering therapy in patients at very high cardiovascular risk. Clin Investig Arterioscler. 2025;37(6):500773. https://doi.org/10.1016/j.arteri.2025.500773
- Mefford MT, Zhou H, Li K, et al. Lipid-lowering therapy utilization, adherence, and outcomes among adults with and at-risk for atherosclerotic cardiovascular disease in an ıntegrated healthcare delivery system in Southern California. Am J Cardiol. 2025;255:55-63. https://doi.org/10.1016/j.amjcard.2025.07.011
- Usherwood T. Therapeutic inertia. Aust Prescr. 2024;47(1):15-19. https://doi.org/10.18773/austprescr.2024.001
Copyright and license
Copyright © 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.